
Edward Schaeffer, MD, PhD
Director, Polsky Urologic Cancer Institute
Edmund Andrews Professor and Chair
Department of Urology
Northwestern University
Feinberg School of Medicine
The Polsky Urologic Cancer Institute of the Robert H. Lurie Comprehensive Cancer Center of Northwestern University at Northwestern Memorial Hospital aims to translate scientific discoveries into meaningful solutions for patients. Our multidisciplinary team of physicians and scientists is dedicated to advancing the quality of urologic cancer care, providing exceptional care across the spectrum of disease, and to creating and maintaining a supportive environment to improve the health and life of our patients.
The Polsky Urologic Cancer Institute owes its creation to a generous gift from Michael and Tanya Polsky. Thanks to the continued support of the Polsky family and other philanthropic partners, the Polsky Urologic Cancer Institute is leading efforts to develop new strategies to diagnose and treat urologic cancers with innovative clinical trials and translational and biotechnological research initiatives.
In partnership with the Lurie Cancer Center, we promote interdisciplinary collaborations to maximize their impact on cancer treatment — and cancer patients — across Northwestern Medicine's expanding health system.
I am pleased to highlight the 2020 accomplishments of our outstanding team of clinicians and scientists. We will continue to build upon this clinical and research excellence to provide the best care for patients through a disease centric program of research, education and discovery.



Leonidas C. Platanias, MD, PhD
Director, Robert H. Lurie Comprehensive Cancer Center of Northwestern University
Jesse, Sara, Andrew, Abigail, Benjamin and Elizabeth Lurie Professor of Oncology
Despite the impact of COVID-19, Lurie Cancer Center remains focused on pushing the boundaries of research and cancer care. Designated as a Comprehensive Cancer Center by the NCI, and the only one in Illinois to receive an "Exceptional" rating, we are leading efforts to translate scientific discovery into better ways to prevent, diagnose and treat cancer.
Our patients benefit from world-class resources, advanced technologies and unrivaled expertise. In addition to targeted therapies and access to cutting-edge treatments through clinical trials, our specialists offer a wide range of services and comprehensive support for patients and families before, during and after cancer treatment.
We thank the Polsky family for their generous support, and Ted Schaeffer for his strong leadership of the Polsky Urologic Cancer Institute of the Lurie Cancer Center at Northwestern Memorial Hospital. Working together across disciplines at Northwestern Medicine, our physicians and scientists are well-positioned to provide the highest level of patient-centered care for all types of urologic cancers.



The Polsky Urologic Cancer Institute of the Lurie Cancer Center provides patients with a multidisciplinary access point for urologic cancer care. Bolstering promising, active research, we offer groundbreaking innovation in the prevention, diagnosis and management of urologic cancers.
 Nationally recognized prostate cancer expert
Ashley Ross, MD, PhD, joined the Polsky Urologic Cancer Institute in July 2020. A surgeon-scientist who specializes in urologic oncology, Dr. Ross is an Associate Professor of Urology at Feinberg and a member of the Lurie Cancer Center. His research efforts focus on the development, testing and implementation of novel diagnostics and therapeutics with a goal of reducing the suffering from prostate cancer. Clinically, Ross performs prostate cancer screening, prostate biopsy (including MRI-fusion biopsy), active surveillance, robotic prostatectomy and ablative therapies of the prostate. Prior to joining Northwestern Medicine, he served as director of the Johns Hopkins Urology Prostate Cancer Program, executive medical director of the Mary Crowley Cancer Research Center and an associate chair of the U.S. Oncology Genitourinary Research Committee.
Nationally recognized prostate cancer expert
Ashley Ross, MD, PhD, joined the Polsky Urologic Cancer Institute in July 2020. A surgeon-scientist who specializes in urologic oncology, Dr. Ross is an Associate Professor of Urology at Feinberg and a member of the Lurie Cancer Center. His research efforts focus on the development, testing and implementation of novel diagnostics and therapeutics with a goal of reducing the suffering from prostate cancer. Clinically, Ross performs prostate cancer screening, prostate biopsy (including MRI-fusion biopsy), active surveillance, robotic prostatectomy and ablative therapies of the prostate. Prior to joining Northwestern Medicine, he served as director of the Johns Hopkins Urology Prostate Cancer Program, executive medical director of the Mary Crowley Cancer Research Center and an associate chair of the U.S. Oncology Genitourinary Research Committee.
In this podcast, Dr. Ross discusses his clinical expertise in focal therapy for the management of prostate cancer. He talks about recent advances, patient selection and future directions for this emerging “middle ground” treatment modality.
The Cancer Genetics Program of the Polsky Urologic Cancer Institute provides genetic counseling for prostate/genitourinary-related hereditary cancer syndromes. The Institute’s genetic counselor works with patients and clinicians to translate genetic information into an individualized care plan for patients and their families.
In this video, Ashley Ross, MD, PhD, delves into the genetics of prostate cancer, describing how genetics can influence cancer development and prognosis, and how genetic testing aids in treatment selection.
One of the greatest challenges in prostate cancer management is identifying patients that will progress to advanced disease following treatment. Our patient-centered genomics research program is working to broaden our understanding of the molecular features of those who do and do not respond to treatment. In the clinical setting, our physicians collaborate with leading genomic laboratories to develop personalized treatment plans based on the genomic profile of a patient’s tumor.
Edward Schaeffer, MD, PhD, is conducting research to identify the molecular mechanisms associated with aggressive prostate cancer. The outcomes of this research will assist in the development of robust biomarkers for treatment intensification and improved clinical outcomes. In this video, he shares details about the clinical application of this work.

The EDGe BRCA+ Men’s Clinic provides screening and follow-up care for men with identified mutations in the BRCA1 and BRCA2 genes. In order to qualify for enrollment, men need to have a known BRCA mutation and be over the age of 40.
The clinic will coordinate 1-2 visits per year across the following specialties:
Established in 2020, the Von Hippel-Lindau (VHL) Clinic at Northwestern Memorial Hospital is one of two Von Hippel-Lindau Syndrome Alliance recognized Clinical Care Centers in Illinois. VHL is a complex hereditary cancer syndrome that requires care across multiple disciplines, including Urology, due to the increased risk of renal cancer. Brittany Szymaniak, PhD, CGC, and Gregory Auffenberg, MD, MS, are leading efforts within the Polsky Urologic Cancer Institute to evaluate and manage patients with VHL syndrome and the associated risk factors.

Brittany Szymaniak, PhD, CGC, leads clinical genetics in the Polsky Urologic Cancer Institute as part of the Northwestern Medicine Cancer Genetics Program. Brittany graduated from the Northwestern University Graduate Program in Genetic Counseling in 2018 and received her PhD in Genetics from the University of Rochester in 2016. Her doctoral work focused on understanding the consequences of loss of ATM on glial precursor cell biology in the neurodegenerative condition, Ataxia-telangiectasia.

Greg Auffenberg, MD, MS, designed and led a quality improvement initiative involving urologists from across the Northwestern Medicine system. The team is piloting the use of a structured, surgical video-peer review process to evaluate surgeon-specific variation in the performance of transurethral resection of a bladder tumor (TURBT). TURBT is the means for diagnosis and initial treatment of the approximately 80,000 bladder cancer cases diagnosed annually in the United States. Although a common procedure — performed five or more times by 35 different surgeons across Northwestern Medicine in the last year — it is technically challenging and quality varies. Future directions of this work include a system-wide surgical coaching intervention focused on improving rates of detrusor sampling across the healthcare system. Capture of detrusor muscle during TURBT has implications for cancer staging and treatment, has been associated with decreased mortality, and may help avoid costly repeat procedures.
The COVID-19 pandemic presented new challenges, concerns and priorities for patients and clinicians, alike. Throughout the pandemic, the Polsky Urologic Cancer Institute offered telehealth and implemented new in-office safety measures to ensure that any individual with a urologic cancer diagnosis had immediate access to clinicians and treatment. We also continued to conduct high-impact research to advance care.
Read about the Lurie Cancer Center's response to the COVID-19 Pandemic »
In these videos, Joshua Meeks, MD, PhD, Alicia Morgans, MD, MPH, and Edward Schaeffer, MD, PhD, discuss recommendations and new clinical protocols in place for patients.

This past year, investigators at the Polsky Urologic Cancer Institute of the Lurie Cancer Center made transformative new insights in the field of urologic oncology. Below are some notable achievements with promising potential to advance knowledge and improve outcomes for urologic cancer patients.

Sarki Abdulkadir, MD, PhD, and his team have discovered a new target for slowing treatment-resistant prostate cancer, according to a recent study published in Nature Communications.
Most prostate cancers are driven by an androgen receptor that can cause downstream upregulation in cell proliferation proteins, such as MYC. This growth can sometimes be kept under control with treatments that reduce androgen, but many cancers have mechanisms that essentially produce their own androgen, eventually rendering such therapies as ineffective.
In the current study, Dr. Abdulkadir found that an epigenetic regulator called DOT1L was upregulated in patients with prostate cancer across several large genetic datasets. Treating human prostate cancer cells with a DOT1L inhibitor suppressed the MYC pathway and blunted growth of cancer. To learn more, the scientists conducted genome transcription and binding analysis to examine what happens when DOT1L is inhibited. They found the region of the genome that codes for MYC also contains sites regulated by both DOT1L and androgen receptors. In turn, MYC represses enzymes that degrade androgen receptors and MYC itself. Thus inhibiting DOT1L triggers a vicious cycle that further decreases MYC and androgen receptor levels.
However, the magnitude of the effect was not the most striking finding. That distinction belongs to the self-targeting nature of DOT1L inhibition. According to Abdulkadir, cells without androgen receptors do not have the unique co-location of DOT1L, androgen receptors and MYC — meaning a DOT1L inhibitor may specifically target prostate cancer cells while leaving other tissues alone.
“DOT1L doesn’t exist just to give people cancer, it has a normal function throughout the body and if you inhibit it, it could cause side effects,” Abdulkadir said. “But some tissues, such as these androgen receptor positive cells, regulate DOT1L and MYC in a unique way. You could possibly use a smaller dose so other tissues are not dramatically affected.”
Abdulkadir and his collaborators have already begun searching for drugs that can inhibit DOT1L in humans, as the inhibitors could benefit patients with treatment-resistant prostate cancer.
Learn more about the role of MYC in prostate cancer treatment »

For the first time, advanced prostate cancer has been treated based on the genomic makeup of the cancer, delaying progression for patients with metastatic castration-resistant prostate cancer.
Published in the New England Journal of Medicine, the large, international phase 3 clinical trial, led by Maha Hussain, MBChB, represents a breakthrough in treating this notoriously deadly cancer and for precision medicine more broadly. “We are now entering a new era of personalized care and precision medicine for metastatic prostate cancer. This will change clinical care for prostate cancer,” said Hussain, who is also deputy director of the Robert H. Lurie Comprehensive Cancer Center of Northwestern University and a member of the Polsky Urologic Cancer Institute Advisory Board.
The trial included two cohorts of patients based on their cancer genetic mutations. The median duration of overall survival in patients in cohort A with mutations on BRCA1, BRCA2 or ATM genes was 19.1 months with olaparib compared to 14.7 months with standard hormone therapy, a number considered clinically and statistically significant.

Metastatic bladder cancer is one of the most aggressive cancers, and female gender is associated with a 20% worse survival rate than is the male gender. Joshua Meeks, MD, PhD, is investigating the gender disparities in bladder cancer by dissecting the tumor and evaluating immune mechanisms of resistance to chemotherapy and immunotherapy. This research may translate into novel pathways and potential therapeutic targets to improve outcomes and reduce gender disparities in bladder cancer.
In this video, Meeks shares details about his work. Watch to learn more »
To complement his laboratory research, Dr. Meeks collaborated with Alicia Morgans, MD, MPH, to conduct a qualitative study to better understand the experiences and needs of female bladder cancer patients. The study explores female patients’ perspectives on diagnosis, treatment, educational and psychosocial resources, survivorship, and the overall care experience. The findings of this research will be applied to improve care for females facing bladder cancer at Northwestern and nationwide.

Edward Schaeffer, MD, PhD, is conducting research to better understand racial disparities in prostate cancer in order to improve identification, treatment selection and outcomes for men of all races with prostate cancer.
Dr. Schaeffer’s recent research suggests that Black men with metastatic prostate cancer were more likely to have tumor mutations than white or Asian men. The findings, published in The New England Journal of Medicine, highlight the significance genetic drivers have in increasing one’s risk for developing aggressive prostate cancer.
“This study provides insight into the differences in the biology of the tumors based on an individual’s race, different genetic mutations that may act as drivers of cancer aggressiveness and, in the future, hopefully result in more precision medicine-based treatment approaches,” said Schaeffer.

With funding from the Polsky Urologic Cancer Institute Research Award, Qi Cao, PhD, developed TADsplimer, the first computational tool to systematically detect topologically associating domain (TAD) splits and mergers across the genome between Hi-C samples. TADsplimer recaptures splits and mergers of TADs with high accuracy in simulation analyses and defines hundreds of TAD splits and mergers between pairs of different cell types, such as endothelial cells and fibroblasts. Published in Genome Biology, this work reveals a key role for TAD remodeling in epigenetic regulation of transcription and delivers the first tool to perform dynamic analysis of TAD splits and mergers in numerous biological and disease models.

Praveen Thumbikat, PhD, is examining the role of immune cells in bone cancer pain. Recent research published in Prostate, showed that mice receiving prostate cancer cells had a significantly lower threshold for pain compared with their control counterparts. This is the first study to examine an immunocompetent model of prostate cancer metastasis of the bone and the neurological contribution to chronic pain. The research suggests that nerve growth factor and p75NTR are involved in chronic pain in bone metastasis. Future research is needed to develop analgesics for cancer-induced bone pain.
As part of the Lurie Cancer Center, a National Cancer Institute (NCI)–designated Comprehensive Cancer Center, the Polsky Urologic Cancer Institute provides leading-edge clinical trials for patients with genitourinary cancers, many of which are only available at Northwestern.

Joshua Meeks, MD, PhD, is the translational medicine primary investigator for this national phase III trial to study how well chemotherapy and radiation therapy work with or without atezolizumab in treating patients with localized muscle invasive bladder cancer. This is the largest trial of bladder preservation (involving chemoradiotherapy) to date, with more than 400 patients randomized to chemoradiotherapy alone or with added systemic immunotherapy (atezolizumab). Given the significant morbidity with bladder removal, Dr. Meeks hopes the outcomes of this trial will establish a new standard for bladder preservation.

Led by David VanderWeele, MD, PhD, this is the first neoadjuvant trial specifically treating patients with high-risk prostate cancer that has lost PTEN expression. Loss of PTEN is one of the most common genetic changes in prostate cancer and is also associated with poor outcomes. This trial is testing a novel drug combination comprised of the AKT inhibitor ipatasertib and the androgen receptor antagonist darolutamide. The addition of ipatasertib to abiraterone recently met one of its primary outcomes in a large phase III trial in patients with castration resistant prostate cancer.

Edward Schaeffer, MD, PhD, is conducting an investigator-initiated trial to test the efficacy of PET/MRI for the staging of newly diagnosed prostate cancer. Dr. Schaeffer and his team hypothesize that the synergism of PET and MRI can offer vastly superior anatomic detail and biologic data that are extremely valuable in advanced evaluation of prostate cancer, particularly with respect to surgical and radiation planning, as well as in assessing therapy response. Dr. Schaeffer is planning to conduct a second trial to assess whether PET/MRI’s increased diagnostic capabilities may be useful in identifying candidates for active surveillance or focal therapies.
Maha Hussain, MD, is conducting an investigator-initiated, multicenter, biomarker-preselected, randomized phase II trial evaluating the role of olaparib and hormone therapy in men with metastatic castration-resistant prostate cancer (mCRPC) and DNA repair mutations. mCRPC is a cancer that has spread beyond the prostate gland, and for which hormone therapy is no longer effective in stopping or slowing the disease. "Metastatic prostate cancer is a complex disease, characterized by interpatient and intrapatient heterogeneity," says Hussain. "Therapy development will need to focus on disease biology and treatment personalization."

Sarki Abdulkadir, MD, PhD, has been named the winner of the 2020 Tripartite Legacy Faculty Prize in Translational Science and Education. Feinberg School of Medicine presents the Tripartite Legacy Faculty Prize annually to a faculty member who has demonstrated excellence in research that emphasizes translational approaches, teaching and mentoring and leadership. In this video, Dr. Abdulkadir describes his work investigating the molecular pathways that drive prostate cancer. Watch to learn more »
William Catalona, MD, was awarded the Ramon Guiteras Award by the American Urological Association for outstanding leadership in demonstrating the value of PSA testing and surgical management of prostate cancer.

Milan Mrksich, PhD, has been named Northwestern University’s Vice President for Research. Dr. Mrksich received a Polsky Urologic Cancer Institute Research award in 2019 to develop a megamolecule-based targeted alpha therapy for prostate cancer.

Qi Cao, PhD, received a Department of Defense Prostate Cancer Research Program Award for his proposal "The Role of BMI-associated IncRNAs in Advanced Prostate Cancer." The project aims to determine how lncRNAs work together with BMI1 proteins to influence prostate cancer tumor growth, drug-resistance and metastasis. Dr. Cao hopes this project will provide a strong rationale to develop therapies targeting BMI1-binding lncRNAs or BMI1-activated lncRNAs to reduce the mortality of prostate cancer.

Shilajit Kundu, MD, received the Lurie Cancer Center's new Population Science Translational Bridge Award for his proposal "Understanding prostate cancer in men with inflammatory bowel disease." This collaborative project will explore the shared underlying genetic basis of inflammatory bowel disease and prostate cancer.

The Veterans Health Administration awarded a Biomedical Laboratory Research and Development Clinician Scientist Investigator Award to Joshua Meeks, MD, PhD, for his proposal “Epigenetic Regulation of Immune Evasion in Bladder Cancer.” This four-year project aims to develop a mode for delivering precision therapy for bladder cancer by identifying the genetic and epigenetic targets in a patient’s tumor.
Adam Murphy, MD, MBA, MSCI, was awarded a 5-year R01 by the National Cancer Institute to create cost effective strategies to identify clinically significant prostate cancer while helping avoid unnecessary biopsies among Black and Hispanic men with elevated PSA.

Edward Schaeffer, MD, PhD, received a 5-year R01 by the National Cancer Institute for his proposal “Evaluation of Transperineal Biopsy under Local Anesthesia: A Novel Approach to Decrease Post-Biopsy Infections and Improve Cancer Detection.” He will collaborate with Jim Hu, MD, MPH, at Weill Cornell Medicine to conduct a multisite randomized controlled trial to evaluate in-office transperineal MRI-targeted prostate biopsy.
The Polsky Urologic Cancer Institute provides emotional and practical support resources tailored to the needs of urologic cancer patients.


We are thrilled to welcome Mandi Buss, LCSW, to the Lurie Cancer Center Supportive Oncology Program at Northwestern Memorial Hospital. Mandi is a licensed clinical social worker who will be providing psychosocial services to urologic cancer patients, their families, and their caregivers.
Mandi has over nine years of diverse experience as a social work clinician, providing psychosocial care to individuals, couples, and families in the context of cancer diagnoses, mental health crises, and other major life disruptions. Her clinical interests include adjustment and coping; personal grief/loss within the individual experience of cancer; and the psychosocial impacts of chronic cancer-related issues.
Mandi previously has worked on pediatric and adult oncology services in Chicagoland and New York City, most recently at Memorial Sloan Kettering Cancer Center where she provided supportive counseling and resource assistance to individuals diagnosed with advanced gastrointestinal cancers.

The Polsky Urologic Cancer Institute hosts disease-specific support groups for bladder and prostate cancers. The groups were moved to a virtual setting due to the COVID-19 pandemic. This expansion fostered connections between the Northwestern Medicine urologic cancer community and urologic cancer patients from around the United States.
Bladder cancer support groups launched in fall 2020. The groups allow participants to connect with other patients, survivors, and caregivers to learn more about bladder cancer and the bladder cancer journey, share experiences and receive support. As a result of research conducted by Drs. Meeks and Morgans, a women’s bladder cancer support group was created to provide a forum for discussion about issues that are unique to women facing bladder cancer. Groups will address female-specific needs such as managing side effects of treatment, impact of bladder cancer on sexual function, and patient-provider communication.
"Updates and Advances in Urologic Oncology" is a monthly didactic lecture series on critical topics in urologic oncology. Presented by the Polsky Urologic Cancer Institute, the series is offered free of charge to any provider involved in the care of patients with urologic cancers.
Directed by Ashley Ross, MD, PhD, the lecture series began in October with Joshua Meeks, MD, PhD, presenting on current issues in non-muscle invasive bladder cancer. In November, Maha Hussain, MBChB, discussed precision medicine for prostate cancer and the use of PARP inhibitors in prostate cancer. Other planned talks include bladder sparing treatment options for muscle invasive bladder cancer, non-radical treatments for prostate cancer, immunotherapy in urologic cancers, and novel prostate biopsies.

Our faculty participated in national committees to develop practice guidelines and clinical recommendations for the management of urologic malignancies.
Jeffrey Sosman, MD, co-authored the National Comprehensive Cancer Network (NCCN) Guidelines for Management of Immunotherapy-Related Toxicities as well as the NCCN Guidelines for Kidney Cancer.
Brittany Szymaniak, PhD, CGC, was first author on “Practical Considerations and Challenges for Germline Genetic Testing in Patients With Prostate Cancer: Recommendations From the Germline Genetics Working Group of the PCCTC.” Published in JCO Oncology Practice, the manuscript describes best practices for integrating genetic counseling and testing for prostate cancer patients into urology clinic workflows.

Lauren Cooley, MD, PhD, a fourth year resident in the Department of Urology, is the recipient of a Urology Care Foundation Residency Research Award. These awards provide motivated and exceptional urology residents with mentored training to prepare them for careers in urologic research. Cooley will work under the direction of Joshua Meeks, MD, PhD, to investigate the immunological and genetic based differences in early and late bladder cancer tumorigenesis between genders. Dr. Cooley also received the 2020 Elisabeth Pickett Research Award by the Society for Women in Urology for her research studying gender disparities in bladder cancer.

Yara Rodriguez, a graduate student working in the Abdulkadir lab, was awarded a highly competitive Association of American University Women International Fellowship award for her research studying the genetic drivers of therapy-resistant prostate cancer.

YongYong Yang, PhD, a postdoctoral fellow in the Cao lab was awarded a Department of Defense Prostate Cancer Research Program Young Investigator Award. Dr. Yang's research aims to elucidate critical insights to prostate progression and identify new effective therapeutic methods for patients with castrate resistant prostate cancer.

Young A. Yoo, PhD, received the Robert J. Krane, MD, Urology Research Award by the American Urological Association. She will be working under the mentorship of Sarki Abdulkadir, MD, PhD, to study androgen deprivation therapy resistance in prostate cancer.
Adam Murphy, MD, MBA, MSCI, is conducting research on prostate cancer, patient education and screening for high-risk patients. He is especially interested in the causes of prostate cancer and the source of poorer health outcomes faced by marginalized groups.
Murphy's team implemented basic infrastructure to integrate urology education into the surgical residency curriculum at the John F. Kennedy Hospital in Monrovia, Liberia, to embark on bidirectional educational exchange and future research endeavors. Through the collaboration and guidance of the Northwestern team, the department at John F. Kennedy Hospital achieved regional accreditation by the West African College of Surgeons for their surgical residency training program.
Dr. Murphy also led a population-based study to evaluate the overall prevalence of prostate cancer in Nigeria as well as to unmask risk factors for aggressive prostate cancer in an unscreened Nigerian population. Public support for Nigerian cancer registries is on the rise, so Dr. Murphy hopes that elucidating the epidemiology of prostate cancer and of aggressive prostate cancer will result in making targeted screening for aggressive prostate cancer a public health priority.
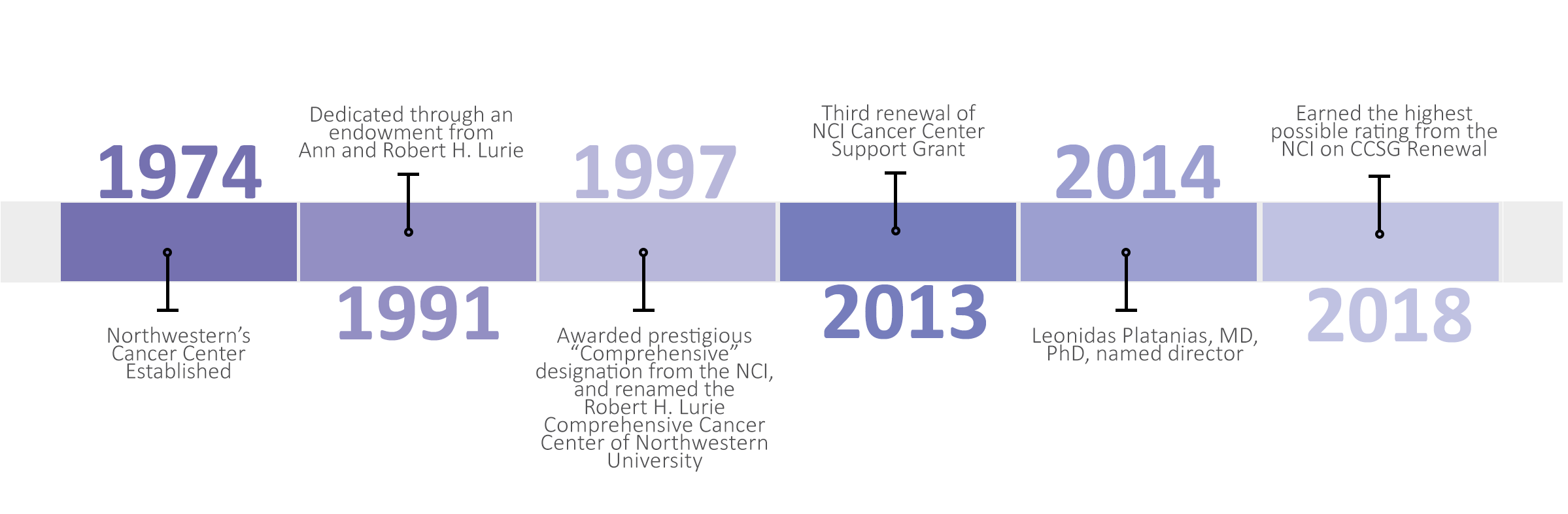
Northwestern’s cancer center, founded in 1974, was dedicated in 1991 through an endowment from Ann and Robert H. Lurie. The title was modified in 1997, when the Lurie Cancer Center received the National Cancer Institute’s (NCI) prestigious “Comprehensive” designation, a reflection of our dedication to the highest standards of cancer research, patient care, education and community outreach.
Lurie Cancer Center is a founding member of the National Comprehensive Cancer Network (NCCN), an alliance of 28 of the world's leading cancer centers, devoted to improving the quality and effectiveness of cancer care and also part of the Big Ten Cancer Research Consortium, a network of academic institutions working together on highly translational clinical trials using the expertise of Big Ten universities.
Through our clinical partnerships with Northwestern Memorial Hospital, Ann & Robert H. Lurie Children’s Hospital of Chicago, Shirley Ryan AbilityLab and the Jesse Brown VA Medical Center, Lurie Cancer Center is uniquely positioned to offer patients leading-edge treatments and exceptional cancer care.

For more information, please visit the
Polsky Urologic Cancer Institute website
Follow Northwestern Urology on Twitter



© Robert H. Lurie Comprehensive Cancer Center
of Northwestern University
cancer.northwestern.edu | cancer@northwestern.edu